Official FDA Manufacturing License
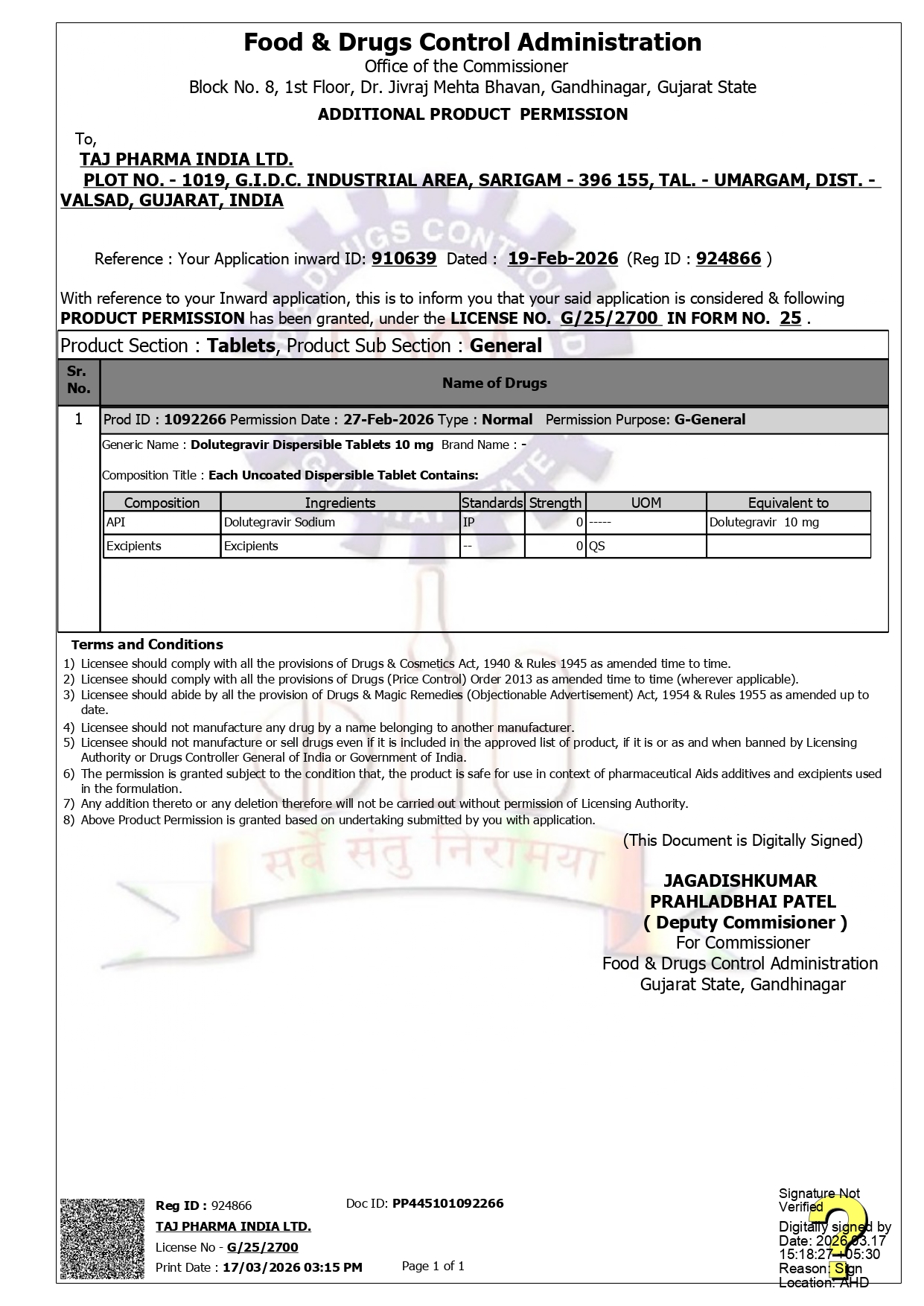
License Details
Product
Dolutegravir 10mg Dispersible Tablets (TAPIVIR DT 10)
Manufacturer
Taj Pharmaceuticals Limited, Vapi, Gujarat
Regulatory Status
FDA Approved Manufacturing License
Distribution
Approved for domestic and export markets
Regulatory Compliance
This FDA Manufacturing License certifies that Taj Pharmaceuticals Limited meets all stringent regulatory requirements for pharmaceutical manufacturing. The license ensures that TAPIVIR DT 10 is manufactured under the highest quality standards and safety protocols, especially for pediatric use.
Key Compliance Areas:
- Good Manufacturing Practice (GMP) standards
- Quality control and testing procedures
- Pediatric safety and efficacy requirements
- Regulatory documentation and record-keeping
- Facility inspection and certification
- Adverse event reporting and monitoring
FDA Approved & Certified
TAPIVIR DT 10 is manufactured under FDA-approved license, ensuring highest quality and safety standards for pediatric patients worldwide.